A COMPREHENSIVE QUANTITATIVE AND QUALITATIVE EVALUATION OF EXTRAPOLATION OF INTRAVENOUS PHARMACOKINETIC PARAMETERS FROM RAT, DOG, AND MONKEY TO HUMANS. I. CLEARANCE
4.5 (281) In stock

This study was conducted to comprehensively survey the available literature on intravenous pharmacokinetic parameters in the rat, dog, monkey, and human, and to compare common methods for extrapolation of clearance, to identify the most appropriate species to use in pharmacokinetic lead optimization, and to ascertain whether adequate prospective measures of predictive success are currently available. One hundred three nonpeptide xenobiotics were identified with intravenous pharmacokinetic data in rat, dog, monkey, and human; both body weight- and hepatic blood flow-based methods were used for scaling of clearance. Allometric scaling approaches, particularly those using data from only two of the preclinical species, were less successful at predicting human clearance than methods based on clearance as a set fraction of liver blood flow from an individual species. Furthermore, commonly used prospective measures of allometric scaling success, including correlation coefficient and allometric exponent, failed to discriminate between successful and failed allometric predictions. In all instances, the monkey tended to provide the most qualitatively and quantitatively accurate predictions of human clearance and also afforded the least biased predictions compared with other species. Additionally, the availability of data from both common nonrodent species (dog and monkey) did not ensure enhanced predictive quality compared with having only monkey data. The observations in this investigation have major implications for pharmacokinetic lead optimization and for prediction of human clearance from in vivo preclinical data and support the continued use of nonhuman primates in preclinical pharmacokinetics.

PDF] A comprehensive quantitative and qualitative evaluation of extrapolation of intravenous pharmacokinetic parameters from rat, dog, and monkey to humans. I. Clearance.

Quantitative Assessment of Blood Lactate in Shock: Measure of Hypoxia or Beneficial Energy Source

Comprehensive Assessment of Human Pharmacokinetic Prediction Based on In Vivo Animal Pharmacokinetic Data, Part 1: Volume of Distribution at Steady State - Lombardo - 2013 - The Journal of Clinical Pharmacology - Wiley Online Library

Preclinical toxicology and safety pharmacology of the first-in-class GADD45β/MKK7 inhibitor and clinical candidate, DTP3. - Abstract - Europe PMC

A COMPREHENSIVE QUANTITATIVE AND QUALITATIVE EVALUATION OF EXTRAPOLATION OF INTRAVENOUS PHARMACOKINETIC PARAMETERS FROM RAT, DOG, AND MONKEY TO HUMANS. I. CLEARANCE
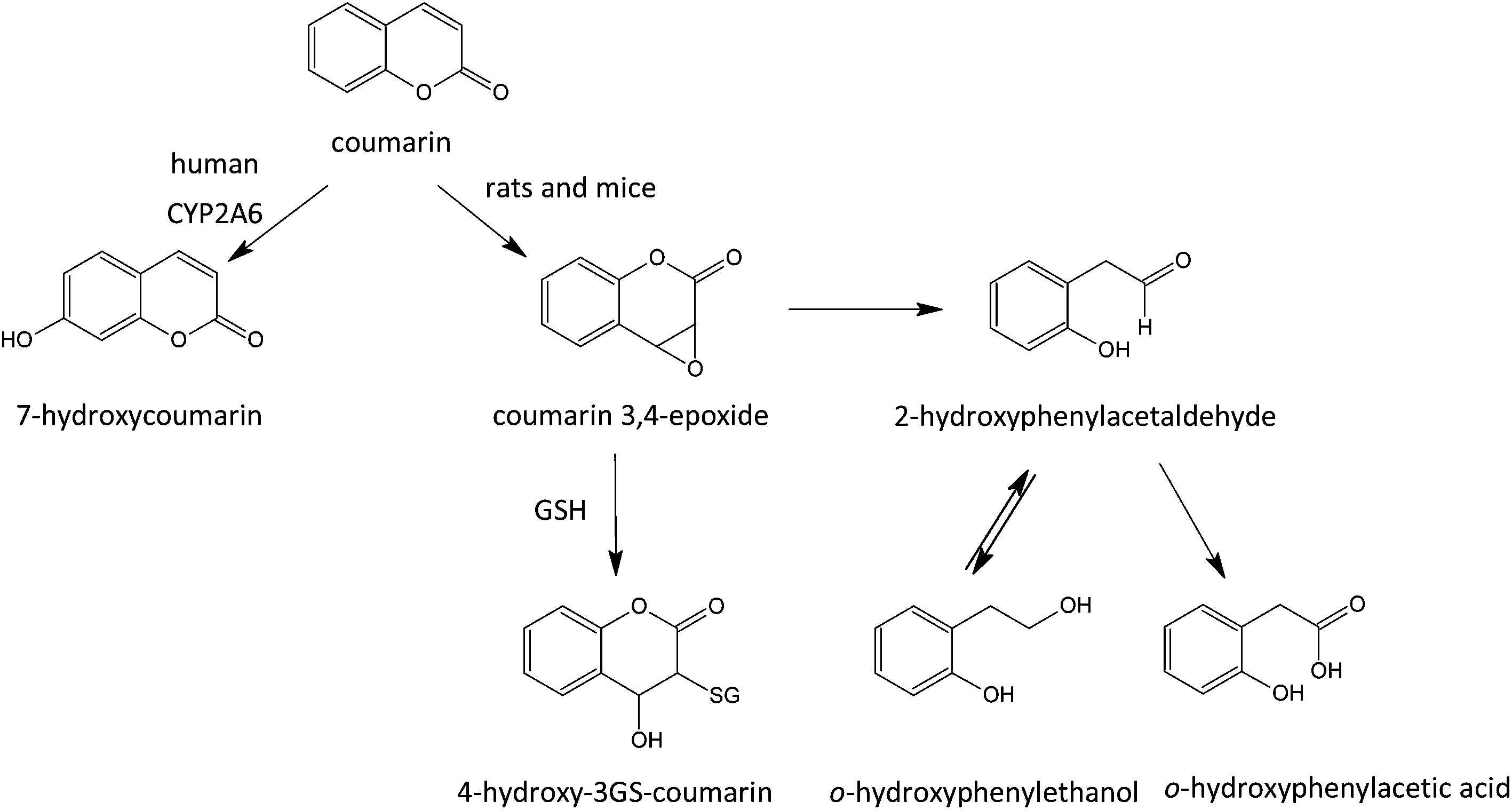
The safety evaluation of food flavouring substances: the role of metabolic studies - Toxicology Research (RSC Publishing) DOI:10.1039/C7TX00254H

Full article: A new approach to predicting human hepatic clearance of CYP3A4 substrates using monkey pharmacokinetic data
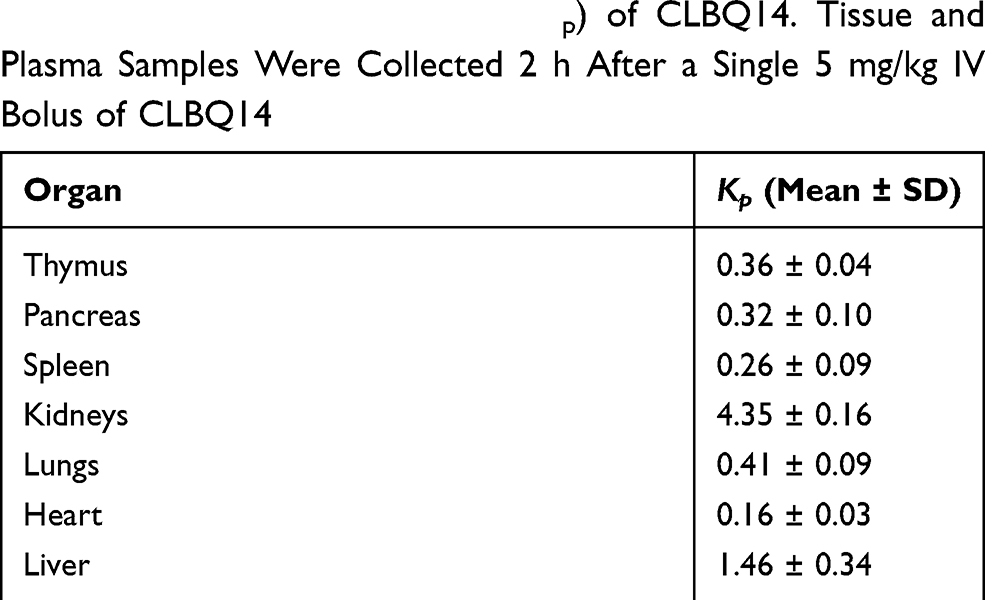
Pre-Clinical Pharmacokinetics, Tissue Distribution and Physicochemical

Pharmacokinetics of a Single Dose of Oral Meloxicam in Rehabilitated Wild Brown Pelicans (Pelecanus occidentalis)

PDF] A comprehensive quantitative and qualitative evaluation of extrapolation of intravenous pharmacokinetic parameters from rat, dog, and monkey to humans. I. Clearance.
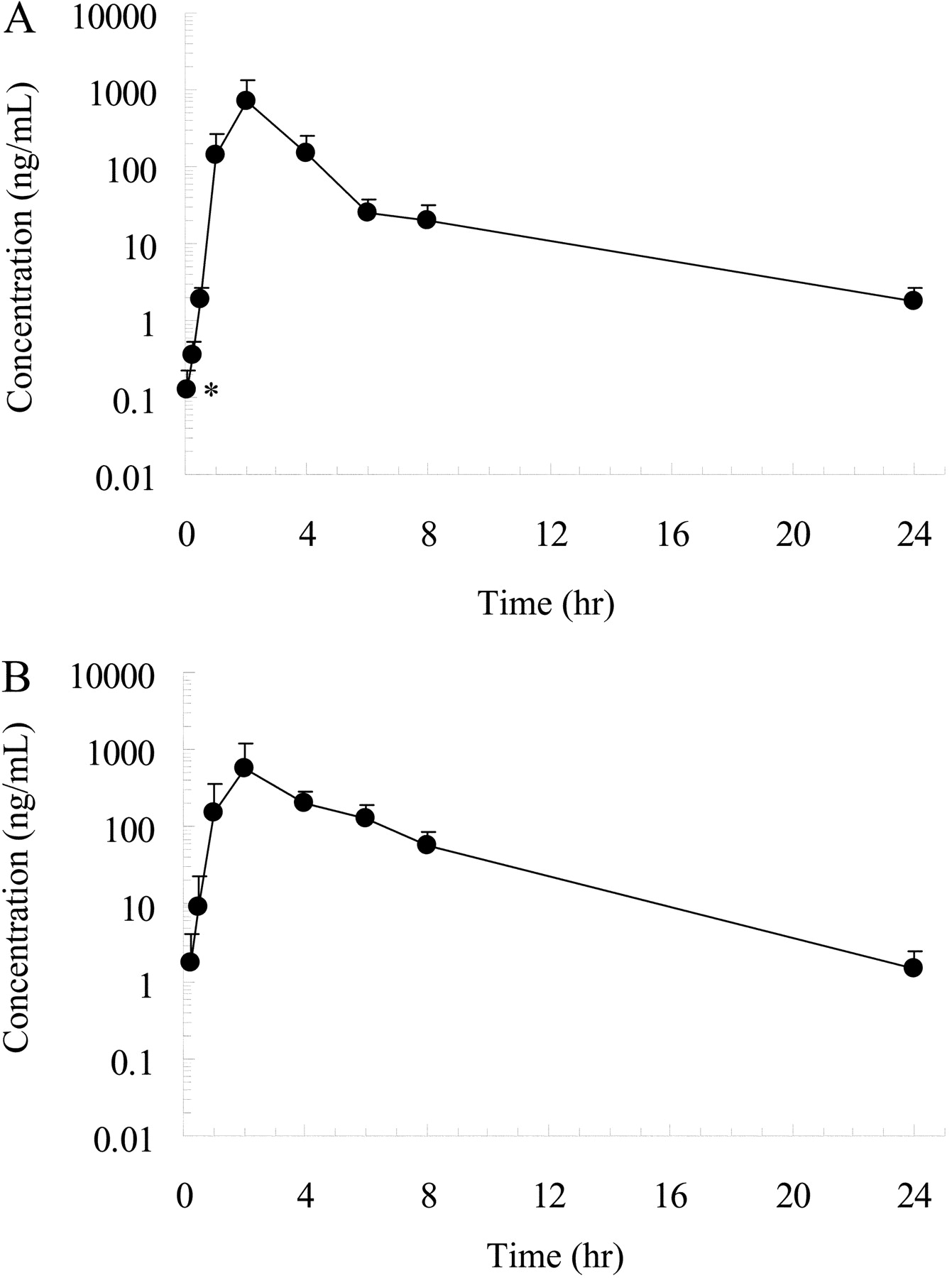
Effect of Oral Ketoconazole on Oral and Intravenous Pharmacokinetics of Simvastatin and Its Acid in Cynomolgus Monkeys
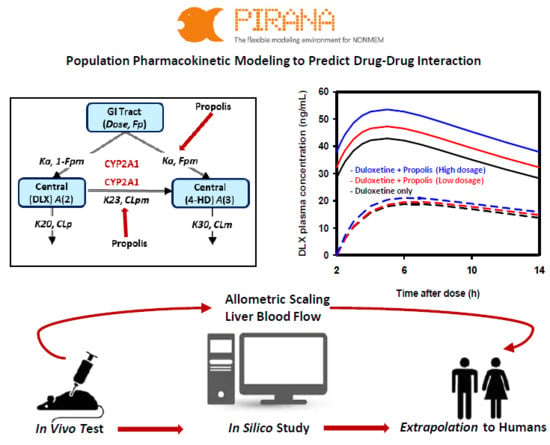
IJMS, Free Full-Text

PDF) Exploration of the African green monkey as a preclinical pharmacokinetic model: Oral pharmacokinetic parameters and drug-drug interactions

High Expression of UGT1A1/1A6 in Monkey Small Intestine: Comparison of Protein Expression Levels of Cytochromes P450, UDP-Glucuronosyltransferases, and Transporters in Small Intestine of Cynomolgus Monkey and Human
Mrat Sequin Sleeve Blouse Clearance Elegant Shirt Long Sleeve
Mrat Winter Coats for Women UK Clearance, Ladies Plush Lining Coat
 Booker Yoga Pants Womens Casual High Waist Loose Solid Color Comfy Stretch Yoga Wide Leg Pants
Booker Yoga Pants Womens Casual High Waist Loose Solid Color Comfy Stretch Yoga Wide Leg Pants Puma x AMI Sweatpants Black
Puma x AMI Sweatpants Black 80s Leg Warmers Set for Women Neon Workout Outfit 80s Costumes Accessories Leg Warmers Running Headband Wristbands for Girls, Blue, Large : : Clothing, Shoes & Accessories
80s Leg Warmers Set for Women Neon Workout Outfit 80s Costumes Accessories Leg Warmers Running Headband Wristbands for Girls, Blue, Large : : Clothing, Shoes & Accessories- Pantalones purple brand disponibles del 30 al 40 en súper oferta 1,290🔥 Disponibles en @matalo_sport_boutiq ( EL INESPRE DE LOS FLOW
- Tênis ON Running CLOUDSWIFT 3.0 Masculino
 Adult Coloured Tights Pantyhose 8 Colours & 2 Sizes for Fancy Dress Costumes
Adult Coloured Tights Pantyhose 8 Colours & 2 Sizes for Fancy Dress Costumes
